In spatial transcriptomics experiments, we discovered that the cryoembedding method of the material determines the integrity of tissue sections. Therefore, we have made improvements for tomato callus material in this regard, thereby obtaining better sectioning results.
Regarding the cryoembedding of the material, we used tomato callus from the same batch of growth to test the embedding effects of two different embedding agents, Sakura OCT and Neg-50. To minimize the degradation of material RNA as much as possible, all embedding steps were carried out on ice. The specific embedding process is as follows:
- Place the embedding agent into a 2 mL centrifuge tube and precool it on ice.
- Place the material into the 2 mL centrifuge tube with precooled embedding agent, and evacuate for 5 minutes to allow the embedding agent to enter the interior of the material.
- After the evacuation ends, centrifuge at 3000 rpm for 5 minutes at 4℃ to remove excess bubbles around the plant.
- Use a 17×17×5 mm embedding box for embedding, adjust the sample position, and place it in the cryoembedding machine to solidify the embedding agent.
- Place the cryoembedded samples in a sealed bag and store at -80℃.
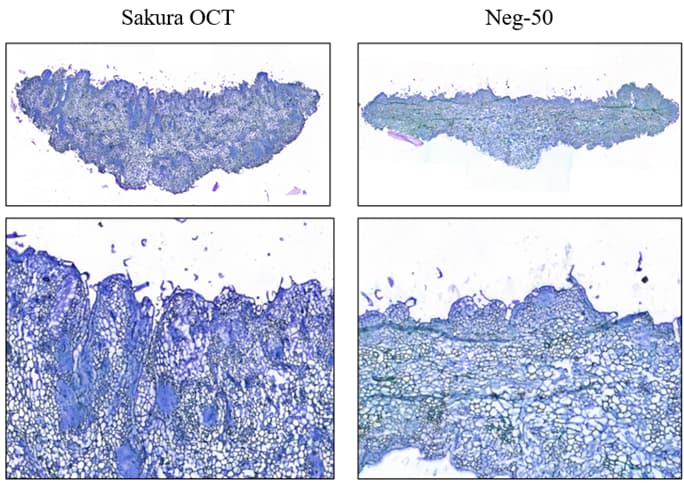
Next, we conducted a test on the sectioning integrity of the cryoembedded samples. The average cell size of tomato callus is 37μm, with smaller cells being around 10μm. To obtain spatial data at the single-cell level, we chose to use a section thickness of 10μm. During the sectioning process, it was found that the sections of callus tissue cryoembedded with Sakura OCT had better integrity, with no obvious fragmentation or shrinkage of the tissue; whereas the sections of callus tissue cryoembedded with Neg-50, although not obviously fragmented, had more folds causing cell stacking, and the stacked cell parts would lead to the disorder of spatial location information. By comparing the sectioning effects of the two embedding agents, we ultimately chose to use Sakura OCT for the cryoembedding of callus tissue.